

|
We are seeing more and more research that shows how critical standard soft lens fitting can be, as it relates to the ultimate success of our patients in
individual contact lenses and even perhaps their long-term wear. Although often taken for granted, soft lens materials and design differ�sometimes quite
dramatically. If you think about it, two contact lenses with the same base curve and diameter can also drastically differ in their material characteristics
(e.g., modulus, oxygen transmissibility, coefficient of friction) in addition to their design (center thickness, edge thickness, edge profile, sagittal
depth). I urge you to take a second look at the fit and contact lens characteristics when things are not working for your patient.
|

|
An American Optometric Association-organized watchdog group will sniff out illegal contact lens sales online and in brick-and-mortar shops. Organized by
the AOA's Contact Lens and Cornea Section (CLCS), the AOA Contact Lens watchdog group will research any violation of federal law and regulations related to
contact lens sales. It also will work with AOA staff to report these incidents and take additional action, in the interest of public health.
Proper contact lens use is a priority for the AOA, which has a campaign to alert consumers of the dangers of purchasing contact lenses without a
prescription. The AOA's 2014 American Eye-Q consumer survey found that 50% of respondents who wore decorative contact lenses purchased them
without a prescription.
The watchdog group will include two arms:
1. A chair in each state who will track sales at gas stations, flea markets, and other venues.
2. An Internet subgroup that will focus on online sales.
Circle lenses, which give the illusion of larger eyes and may emerge as a trend in 2015, offer an example of the types of activities the watchdog group may
be tracking in the coming year.
Anyone who suspects illegal contact lens sale activity can report these retailers to: stopillegalCLs@aoa.org.
These reports will allow for improved tracking of illegal sales and adverse events, says CLCS Chairman Thomas Quinn, OD, and also allow data to be compiled
that can be presented to government agencies, highlighting the problem of illegal contact lens sales.
For more information, contact Kara Webb, AOA associate director for coding and regulatory policy, at kcwebb@aoa.org.
The AOA also has information for reporting illegal contact lens sales available here.
|
|
Bausch + Lomb announced that Vision Source has selected the company as an elite vendor in the therapeutics category and a vendor in contact lenses and lens
care products. As part of the relationship, Vision Source will endorse B+L�s industry-leading products together with member pricing discounts and rebate
programs.
The therapeutics products include Ocuvite, PreserVision, Soothe and Alaway. For skincare, the ELASTIderm eye product portfolio will
be available. In addition to the therapeutics, Vision Source members will have access to the Bausch + Lomb ULTRA, Biotrue ONEday and PureVision brands of
contact lenses. Lens care products include Biotrue and renu multi-purpose solutions, as well as PeroxiClear and the Boston solution line.
|
|
A recent study looked at the aerodynamic benefits of eyelashes and their role in protecting the eye. Measurements of the eyelashes in 22 species of mammals
showed them to possess eyelashes of a length one-third the eye width. Furthermore, wind tunnel experiments confirm that this optimal eyelash length reduces
both deposition of airborne particles and evaporation of the tear film by a factor of two. But eyelashes can be too long or too short for optimum benefits.
The study can be viewed on the Journal of the Royal Society Interface: http://rsif.royalsocietypublishing.org/content/12/105/20141294.
|

|
|

|
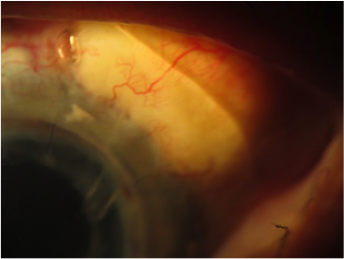
A patient with aphakia, corneal transplant, and bleb was fit with the EyePrintPRO device to vault the bleb and protect the cornea. Her vision went from
hand motion to 20/50.
We thank Dr. Woo for this image and we welcome photo submissions from our other readers! It is easy to submit a photo for consideration for publishing in Contact lenses Today. Simply visit http://www.cltoday.com/upload/upload.aspx to upload
your image. Please include an explanation of the photo and your full name, degree or title and city/state/country.
^ Back to top
|

|
Bacterial Contamination Risk in Cataract Surgery
A recent study from China assessed the bacterial contamination risk in cataract surgery associated with mechanical compression of the lid margin
immediately after sterilization of the ocular surface.
Patients with age-related cataract were randomly assigned to 1 of 2 groups. In Group A (153 eyes), the lid margin was compressed and scrubbed for 360
degrees 5 times with a dry sterile cotton-tipped applicator immediately after ocular sterilization and before povidone-iodine irrigation of the
conjunctival sac. Group B (153 eyes) had identical sterilization but no lid scrubbing. Samples from the lid margin, liquid in the collecting bag, and
aqueous humor were collected for bacterial culture. Primary outcome measures included the rate of positive bacterial culture for the above samples. The
species of bacteria isolated were recorded. Group A and Group B, each comprised 153 eyes. The positive rate of lid margin cultures was 54.24%. The positive
rate of cultures for liquid in the collecting bag was significantly higher in Group A (23.53%) than in Group B (9.80%) (P=.001).The bacterial species
cultured from the collecting bag in Group B were the same as those from the lid margin in Group A. The positive culture rate of aqueous humor in both
groups was 0%.
The authors concluded that mechanical compression of the lid margin immediately before and during cataract surgery increased the risk for bacterial
contamination of the surgical field, perhaps due to secretions from the lid margin glands. What do these results mean for our non-surgical patients for
whom we recommend daily lid hygiene and lid massage, especially those with �compromised� ocular surfaces (dry eye/corneal or conjunctival staining/CL
wearers/trabeculectomy patients etc.)? I submit that we use a product with known antimicrobial activity as opposed to home-made solutions (for example
shampoo/soap based concoctions) to address the presumed increased microbial load from hygiene-related eyelid manipulation.
Ye T, Chen W, Congdon N, Liu Y. Increase in microbial contamination risk with compression of the lid margin in eyes having cataract surgery. J Cataract Refract Surg. 2014 Aug;40(8):1377-81.
^ Back to top
|

|

|
 |
RESEARCH REVIEW
S. Barry Eiden, OD, FAAO
|

|

|

|

|
|
Adverse Events with Daily Disposable CLs � Hydrogel vs. SiHy�
Are we there yet?
A large post market survey study was conducted and reports of adverse events were evaluated. The authors reported annualized adverse events (AEs) including
corneal infiltrative events (CIEs) with use of silicone hydrogel (SiHyDD) and hydrogel daily disposable (HydDD) soft contact lenses (SCLs) in the 1�Day
Acuvue TruEye or 1�Day Acuvue Moist Performance Overview (TEMPO) Registry (NCT01467557).
There were 3064 surveys from 1171 subjects (601 SiHyDD and 570 HydrogelDD) analyzed and calculated for annualized incidence rates of symptomatic AEs during
a 1 year period of time. Three independent experts adjudicated potential AE cases. Demographics were compared between wearers with and without AEs.
The registry tracked 960.3 years of lens wear: SiHyDD 489.4 years and HydDD 470.9 years. In that period, the 601 SiHyDD wearers reported eight AEs with
office visits (1.6%/y; 2 CIEs, 0.4%/y), eight (1.6%/y) without office visits, and four AEs unrelated to SCLs (0.8%/y) (SiHyDD wearers with AEs; 44.8 � 12.5
years; 75% female). The 570 HyDD wearers reported three AEs with office visits (0.6%/y; no CIEs), five without office visits (1.1%/y), and one
non-SCL-related AE (0.2%/y) (HydDD wearers with AEs; 26.3 � 8.0 years; 100% female). These CIE rates are significantly lower than the lowest estimate of
3.3% from prior studies. Wearers with SiHyDD-related AEs were significantly older than unaffected wearers (P = 0.02), but not for HydDD-related AEs.
The authors stated that the CIE rates of 0.4% (SiHyDD) and 0% (HyDD) with these DD lenses are significantly lower than rates previously reported with
reusable SCLs (3%-4%/y), indicating improved safety outcomes with these DD lenses.
So it seems, at least that based upon the evaluation of these two specific DD lens designs, that DD CLs have lower rates of corneal inflammatory events
than with reusable SCLs. Interestingly enough we see lower AE rates with hydrogel DDs vs. SiHy DDs. In fact in the hydrogel DDs the rate of CEIs was zero!
So the take home may be that yes DDs do reduce AE rates when compared to reusable SCLs but that we still have work to do in order to bring the obvious
advantages of high oxygen permeability of SiHy lenses to DD CLs. We are already seeing further development of SiHy DD materials that will bring all of the
optimal performance qualities to this lens wearing modality in order to maximize vision, comfort and eye health response � the three pillars of contact
lens success.
Chalmers RL, Hickson-Curran SB, Keay L, Gleason WJ, Albright R. Rates of Adverse Events With Hydrogel and Silicone Hydrogel Daily Disposable Lenses in a
Large Post Market Surveillance Registry: The TEMPO Registry. Invest Ophthalmol Vis Sci. 2015 Jan 8;56(1):654-63.
^ Back to top
|

|

|
|
Sagittal Height Differences of Frequent Replacement Silicone Hydrogel Contact lenses
|
|
This study aims to evaluate the sagittal height differences among a selection of commercially available monthly or two weekly replacement silicone hydrogel
soft lenses.
The sagittal height (CL-SAG) of four frequent replacement silicone hydrogel lenses (lotrafilcon B, balafilcon A, comfilcon A and senofilcon A) was measured
for all base curve radius manufactured in spherical (-3.00D and +3.00D) and toric (=C-0.75�180�) designs (11 spherical and 8 toric lenses in total). Two
different lenses of each lens type were evaluated (the intra CL-SAG) using the SHSOphthalmic omniSpect by Optocraft.
The intra CL-SAG difference (the difference between two identical lenses from the same batch) was 11�SD 2 μm. The CL-SAG of all minus lenses ranged from
3454 to 3765 μm (an inter CL-SAG difference of 311 μm), while the plus spherical lenses ranged from 3493 to 3757 μm (inter CL-SAG difference 264 μm). In the
toric lens group, the range in toric minus lenses was 3495-3953 μm (inter CL-SAG difference 458 μm) and 3493-3980 μm in the plus group (inter CL-SAG
difference 487 μm). The inter CL-SAG difference between the spherical and the toric lenses was statistically significant (p=0.03).
Marked differences in sagittal height among different commercially available frequent replacement silicone hydrogel lenses exist. Different lenses with the
same package base curve value had marked differences in CL-SAG, with potential clinical significance on-eye in terms of lens behavior. The inter CL-SAG
variance in the spherical lens group was smaller than in the toric lens group.
van der Worp E, Mertz C .Sagittal height differences of frequent replacement silicone hydrogel contact lenses. Cont Lens Anterior Eye. 2015 Feb
18. [Epub ahead of print]
|
|
|

|
|
A Proud Supporter of

Important Links:
To report adverse contact lens reactions visit:
http://www.accessdata.fda.gov/scripts/medwatch/ or call (800) FDA-1088.
To report possible grievances related to the Fairness to Contact Lens Consumers
Act or associated Contact Lens Rule visit:
https://www.ftccomplaintassistant.gov/.
CLToday Services:
Subscribe;
Unsubscribe; Submit Clinical
Image
Submit news to cltoday@pentavisionmedia.com.
Send your comments and fitting tips to
cltoday@pentavisionmedia.com. Please include your full name, degree or title
and city/state/country.
For more information on Contact Lenses Today including archives of previous
issues, please visit our website at www.cltoday.com.
For the latest articles on contact lenses, important clinical information and helpful
tools related to the contact lens practice visit the Contact Lens Spectrum
website at www.clspectrum.com.
© 2015 All Rights Reserved Contact Lenses Today and CLToday
are registered trademarks of:
PentaVision LLC, 321 Norristown Road, Suite 150, Ambler, PA 19002 | 215-628-6550
© 2015 PentaVision LLC All Rights Reserved.
|

|
|
|



















